Platomics announces new collaborations Read the Press Releases

Understanding IVDR IH-IVD Scenarios: A Practical Guide for Laboratories
With the implementation of the In Vitro Diagnostic Regulation (IVDR), laboratories across Europe are facing new regulatory expectations. One of the most important areas concerns in-house IVDs, a concept that allows health institutions to develop or modify diagnostic tests internally under specific regulatory conditions.
While this flexibility enables laboratories to meet unique clinical needs, it also introduces new responsibilities. Understanding when a diagnostic workflow qualifies as an in-house IVD is essential for maintaining compliance. In this article, we explain what in-house IVDs are, explore the five common regulatory scenarios, and outline how laboratories can approach compliance in a practical way.
What Is an In-House IVD?
An in-house IVD is a diagnostic test that is both manufactured and used within the same health institution. These tests are often developed when a suitable CE-marked alternative is not available on the market, or when commercial products do not sufficiently address the clinical needs of specific patients.
Under the IVDR, these internally developed diagnostics fall under Article 5(5). While laboratories are not subject to the exact same regulatory pathway as commercial manufacturers, they are still required to demonstrate that their diagnostic systems are safe, effective, and appropriately documented. This includes complying with the General Safety and Performance Requirements (GSPR) and operating within a quality management system such as ISO 15189, which is widely considered the gold standard for medical laboratories.
Unlike commercial IVDs, where notified bodies are involved in conformity assessment, in-house IVDs are overseen by national competent authorities within EU member states.
The Five IVDR Scenarios
To help laboratories determine when Article 5(5) applies, regulatory experts often distinguish between five practical scenarios. These scenarios represent different levels of modification or development within a diagnostic workflow. They range from scenario 1 with simply using CE-marked devices as intended by the manufacturer which does not qualify as an IH-IVD, up to scenario 5 where a diagnostic system is fully developed from scratch by the laboratory.
LABORATORY TEST ASSESSMENT
Scenario Wizard
Not sure if your test is an In-House IVD according to IVDR?
Let’s find out! By answering a few short questions, our Scenario Wizard helps you determine the status of your test, assign it to the right scenario, and guides you to relevant recommendations.
Scenario 1: Using CE-Marked IVDs Exactly as Intended
The first scenario represents the standard diagnostic setup in many laboratories. Here, the laboratory uses a CE-marked IVD exactly as described by the manufacturer, without modifying any part of the intended use or workflow.
In practice, this means that every aspect of the test is performed according to the manufacturer’s instructions, including how the device is applied, which sample types are used, how the instrument is maintained, and how results are interpreted. Laboratories have to still integrate the device into their internal quality management systems, but they do not alter the fundamental design or intended purpose of the product.
A typical example would be a molecular diagnostics laboratory performing PCR testing for SARS-CoV-2 with a CE-certified kit. The laboratory uses a CE-marked PCR kit, follows the exact protocol described in the instructions for use, runs the test on a compatible thermocycler, and performs quality control with reagents prescribed by the manufacturer. The workflow aligns fully with the manufacturer’s specifications.
This situation means that the regulatory responsibility for safety and performance of the device remains with the manufacturer. The laboratory is responsible for correctly implementing and operating the device in its environment, ensuring staff training, quality control, and proper handling, but it does not need to generate additional technical documentation for the device itself under Article 5(5).
Scenario 2: Off-Label Use of CE-Marked Devices
The second scenario occurs when a laboratory uses a CE-marked device but deviates from the manufacturer’s intended purpose. Even relatively small modifications can move a workflow into the in-house IVD category.
Such deviations may involve using a test with a different type of sample, applying it to a patient group that was not originally specified, or adjusting analytical parameters that influence how the test performs.
For example, a laboratory may decide to apply a PCR test to a specimen matrix that is not listed in the manufacturer’s documentation. The test may have been validated for nasopharyngeal swabs, but the laboratory wants to use it for saliva samples. Another scenario might involve extending the clinical indication of a test to screen for additional conditions beyond the original diagnostic purpose.
Although the device itself has not been physically modified, the laboratory has changed how the test is used and what clinical conclusions are drawn from it.
This situation means that the laboratory assumes additional regulatory responsibility. Because the device is now used outside its validated intended use, the laboratory must demonstrate that the modified workflow still performs safely and reliably. This usually requires additional validation work, performance testing, and documentation to ensure that the change does not negatively affect diagnostic accuracy.
Scenario 3: Using Research-Use-Only Products for Diagnostics
A third common situation arises when laboratories use Research Use Only (RUO) products in diagnostic workflows. RUO products are designed for laboratory research and are not intended for clinical decision-making.
However, in specialized clinical settings laboratories sometimes incorporate RUO products into diagnostic procedures, especially when no commercial alternatives exist.
For instance, a genetic diagnostics laboratory might use an RUO sequencing panel designed for exploratory research to detect rare genetic mutations in patients. The product itself may provide the technical capability to detect these variants, but the manufacturer has not validated it for clinical diagnostic use.
Another example can be found in specialized oncology or immunology laboratories where research reagents are used to detect biomarkers that are not yet covered by commercially approved diagnostic kits.
In these cases, the laboratory effectively changes the intended purpose of the product from research to clinical diagnostics.
This means that the health institution becomes responsible for ensuring the safety and reliability of the diagnostic workflow. Because the original manufacturer did not validate the product for medical use, the laboratory must perform its own validation studies, risk assessments, and ensure the safety of this product as well as documentation to demonstrate that the test is suitable for patient diagnostics.
Scenario 4: Combining Devices Into a New Diagnostic System
Modern laboratory workflows often involve multiple instruments, reagents, and software tools that work together as part of a larger diagnostic system. When these components are combined in ways that were not originally intended by their manufacturers, the resulting workflow may qualify as an in-house IVD system.
A practical example could be a genomic diagnostics pipeline that combines a CE-marked library preparation kit, a sequencing instrument from another manufacturer, and open-source software used to analyze sequencing data. Each component may function correctly on its own, but the manufacturer may not have intended them to be used together in this specific configuration.
Another situation could involve connecting a laboratory instrument to custom-built analysis software that automatically interprets diagnostic results. Even if both elements function individually, their integration creates a new diagnostic system.
This means that the laboratory must demonstrate that the entire workflow works safely and reliably as a combined system. Validation must therefore evaluate not only individual devices but also the interactions between them. Interfaces, compatibility, and data transfer processes must be carefully assessed to ensure that the system consistently produces reliable diagnostic results.
Scenario 5: Fully Developed In-House Diagnostics
The final scenario represents the most comprehensive form of in-house diagnostics: a laboratory develops a diagnostic test entirely on its own.
This situation is particularly common in highly specialized fields such as rare disease diagnostics, advanced genomic testing, or academic medical centers where research-driven innovations are rapidly translated into clinical workflows.
For example, a laboratory may develop a new ELISA assay using raw biological materials to detect a novel biomarker associated with a specific disease. Another laboratory may design a bioinformatics pipeline that processes genomic sequencing data and automatically identifies clinically relevant variants.
Even relatively simple tools can fall into this category. A laboratory might create custom algorithms or spreadsheet-based calculations that interpret laboratory measurements and generate diagnostic results.
In these cases, the laboratory is responsible for the entire lifecycle of the diagnostic system.
This means the health institution effectively takes on a role similar to that of a commercial IVD manufacturer. The laboratory must ensure that the device is properly designed, validated, and documented. Risk management processes, performance evaluations, and quality management procedures must be applied to demonstrate that the diagnostic solution is safe and effective for patient use.
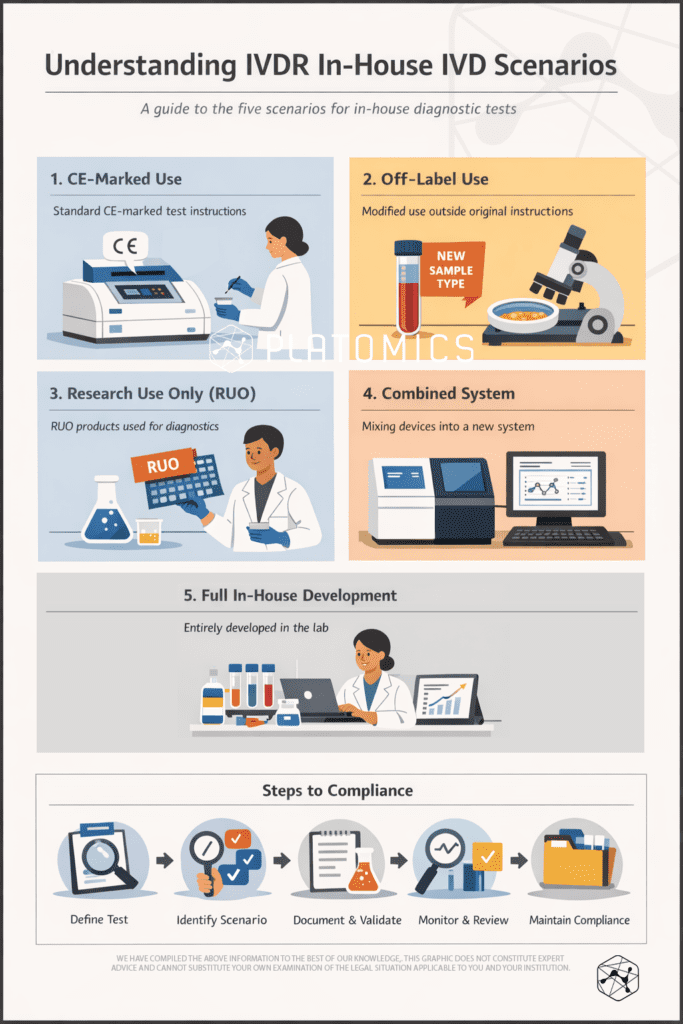
Approaching IVDR Compliance in Practice
Real laboratory workflows rarely fall neatly into a single scenario. In many cases, diagnostic processes combine elements from multiple categories. A workflow might use CE-marked devices, incorporate research reagents, and include internally developed analysis software.
Because of this complexity, laboratories benefit from taking a structured approach to IVDR compliance.
The first step is to clearly define the diagnostic system and identify all components involved. From there, laboratories can determine whether modifications, repurposing, or system integrations introduce in-house IVD requirements. Once the relevant scenario is identified, appropriate validation activities and documentation can be implemented in a risk-based and structured way.
Ongoing monitoring is also important. As workflows evolve, laboratories must ensure that regulatory requirements continue to be met.
Supporting Laboratories in the IVDR Era
Managing in-house IVD compliance can be challenging, particularly for laboratories with complex diagnostic workflows.
Digital tools and structured documentation systems can help laboratories organize regulatory information, maintain traceability, and ensure that compliance requirements are addressed efficiently. By combining regulatory expertise with automation, laboratories can reduce administrative burden while maintaining high quality standards.
At Platomics, the goal is to support laboratories throughout this process by providing software solutions and educational resources designed specifically for IVDR in-house diagnostics.
For laboratories navigating the in-house device landscape, understanding the IH-IVD Scenarios is essential. IVDR ensures that the devices behind those procedures are safe, effective, and compliant. Want to know more about how PlatoX can help you to achieve compliance? Book a free demo with us!
Still navigating IVDR Challenges?
Understanding how different IVDR scenarios impact your diagnostics strategy can be complex — but the right knowledge makes all the difference!
In this PlatoX Academy webinar, our experts walk you through practical IVDR scenarios, explaining what they mean for manufacturers, laboratories, and compliance teams. Learn how to anticipate regulatory pathways and make informed decisions with confidence.
Watch the webinar now in the PlatoX Academy and stay ahead of IVDR requirements.
practical regulatory training
PlatoX® Academy
*We have compiled the above information to the best of our knowledge, yet our blog entries do not constitute expert advice and cannot substitute your own examination of the legal situation applicable to you and your institution.



